8.6: Generic protocol for the evaluation of apheresis equipment
This protocol sets out the minimum requirements for new apheresis equipment and, in a generic form, the mechanism for assessing acceptability of the equipment hardware, the software and the associated apheresis sets. The specific validation or trial of apheresis collections from new equipment is covered in section 8.7. Novel components, as defined in section 8.1, produced as a result of new equipment will be assessed as detailed in other sections of this chapter.
8.6.1: Minimum requirements
8.6.1.1: General
Equipment should be CE/UKCA/UKNI marked or the Blood Service should participate to facilitate CE/UKCA/UKNI marking.
Manufacturers must comply with Good Automated Manufacturing Practice (GAMP).2
8.6.1.2: Equipment hardware
Equipment should contain the following:
- manual override system
- blood flow monitor
- in-line air detector
- integral blood filter
- anticoagulant flow indicator
- collection volume preset device
- visual audible alarm for procedure completion
- automatic standby mode for power failure
- power up self-check to include all critical safety and operational procedures.
8.6.1.3: Equipment hardware
Software should provide parameters:
- for accepted total blood volume calculation algorithm
- for accepted citrate reinfusion rate calculation algorithm
- for fixed upper limit citrate reinfusion (see Chapter 5)
- for programmable upper limit total collection volume
- must not exceed predetermined fluid reinfusion limits (e.g. citrate, saline)
- for alarm and prevent use of incorrect set (incongruent) for programmed procedure
- prevent procedure where predicted post-collection parameters fall outside programmable safety limits as defined in Chapter 5.
For other measures, see Chapter 5.
8.6.1.4: Apheresis sets
Apheresis sets should have:
- a closed system
- a visual system to minimise risk of transposition of fluid lines
- a microbial filter on ‘spiked’ lines (unless the set will only be used to collect plasma for fractionation)
- a diversion line and pouch for sampling
- a means of preventing incorrect connections to the set for IV fluid (e.g. saline) and anticoagulant.
Consideration should be given to the incorporation of a pouch on the final pack to facilitate bacterial contamination testing.
For other measures, see Chapter 5.
The overall mechanism for equipment acceptance is given in Figure 8.1 for reference. Validation, installation qualification, operational qualification and performance qualification would be defined by the Blood Service, taking account of the advice within these guidelines.
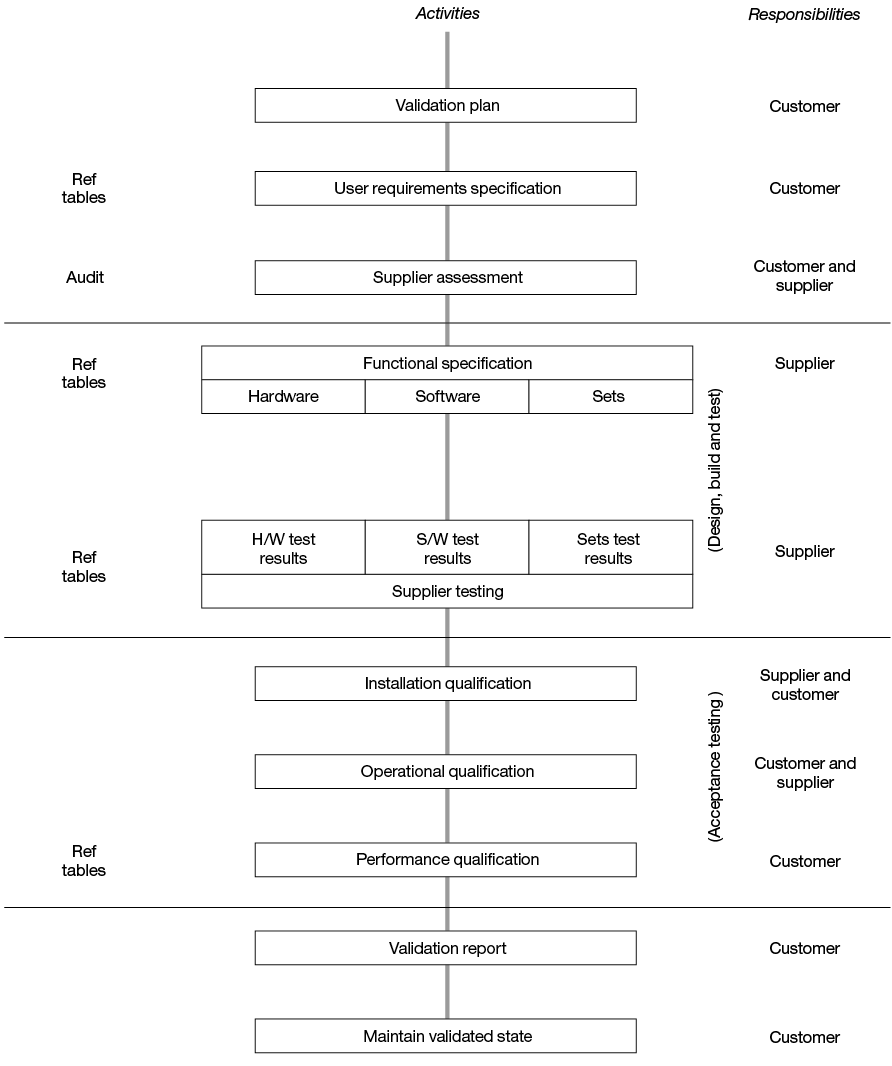
Figure 8.1 Generic flowchart of apheresis equipment acceptance